My 11-year-old son is passionate about all things science.
Since he was very young, he has loved exploring the natural world whether that meant reading about obscure animals, examining funny-looking rocks, or checking the accuracy of the weather forecast.
But as he grows older, he is more drawn to video games, YouTube videos, and apps. And while some of his screen time is educational in nature, recently, I couldn’t help but realize what he needed was more hands-on activities. He needed a chance to learn by doing.
Science kit subscriptions have proven an excellent solution—and MEL Science kits in particular, have been loads of fun.
Along with helping us battle boredom during long weekends at home, they have opened up the world of science, technology, engineering, and math for my children.
A science subscription box is easy, convenient, educational, and, best of all, fun. One of our favorite science kit subscription companies is MEL Science.
Let’s face it: Before we pay anything, we all want to know if we’re getting the best science kits for our kids.
Below, I’ve compiled a guide to let you know what to expect from these boxes, zooming in on the MEL Science Chemistry subscription box in particular.
What is MEL Science?
MEL Science is a monthly science kit subscription service that delivers science experiments to your doorstep once per month.
They have several different lines for children ranging from ages 5 to 14+. Each line has a different scientific focus.
What Do MEL’s Science Subscription Boxes Offer?
Each box contains the materials, equipment, and instructions necessary to enjoy up to three experiments.
The activities are designed to make science fun and interesting for children.
The boxes are organized by age and created with both education and safety in mind.
Depending on the box, and the child’s age, an adult might watch or help out a bit.
MEL Science Kits
Here’s a rundown of the box types MEL Science offers:

MEL Chemistry
The MEL Chemistry subscription is suited for children aged 10 and up.
The first month’s box is a starter kit and two experiments, and a VR headset. Then, each month, two to three new chemistry experiments arrive.
Experiments include:
- Turning cool liquid into hot ice.
- Growing copper coral on an iron clip.
- Causing a zinc pellet to sprout metallic quills.
In my opinion, this may well be the best chemistry set for 11-year-olds.
We’ll take an in-depth look at the MEL Chemistry box below where we unpack the experiments and try them out.

MEL Kids
The MEL Kids subscription is geared to ages 5 to 10+.
Each box features an in-depth project and also comes with AR lessons and live science classes.
Projects include:
- A robotic hand
- A gyroscope
- DIY headphones
You can learn more about the MEL Science Kids Box in this MEL Kids review, where we also unpack the activities and give them a go: MEL Kids vs. Spangler Science Boxes.
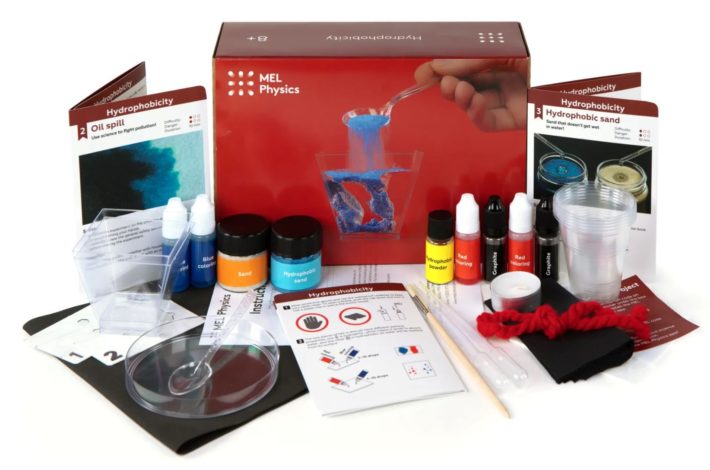
Mel Physics
The MEL Physics subscription is designed for ages 8 to 14+.
Each month’s box contains two to three physics experiments, covering concepts such as internal stress and luminescent lighting.
You can learn more about the MEL Physics box and what’s inside in our MEL Physics box review here: KiwiCo vs. MEL Science Physics.
Pros and Cons of MEL Chemistry Subscription Box
This review will look at the MEL Chemistry Set. Here’s a quick summary of the pros and cons.
MEL Science Special Offers
Pay for 9 months, get 12 months of science: MEL Science
What Was Inside Our MEL Chemistry Science Kit?
Every MEL Science subscription box includes all the equipment, materials, and instructions necessary to complete the activities.
The MEL Chemistry starter kit offers an array of equipment, all packed neatly in a protective foam organizer.
Contents include:
- A crack-resistant Borosilicate glass beaker and flask.
- A macro lens.
- A smartphone stand.
- A fuel burner.
- Two pairs of safety goggles.
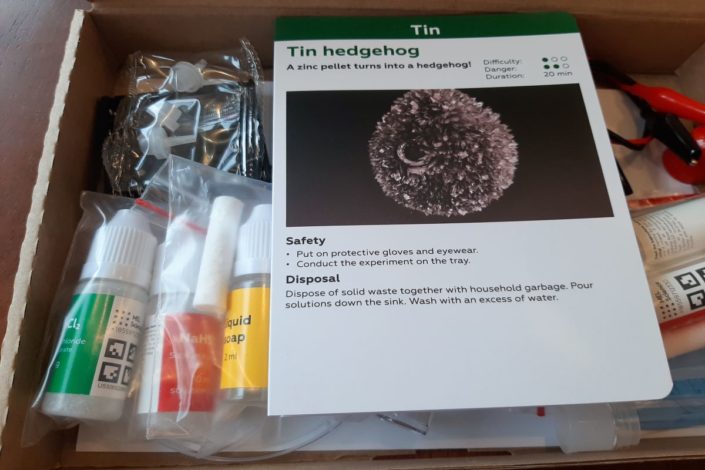
Along with the starter kit, the first MEL chemistry set came with a kit featuring two experiments related to tin: creating a tin “hedgehog” and tin dendrite (which look like a metal “tree”).
The set contained instructions, experiment cards, chemicals, liquid soap, zinc pellets, plastic vials, Petri dishes, pin openers, battery holders, crocodile clip wires, and protective gloves.
We had to supply our own batteries—in this case, four AAA batteries that we happened to have on hand.
How Much Does Mel Science Cost?
The monthly cost for the MEL Chemistry subscription box is $34.90. The starter kit, VR headset, and VR lessons are included at no extra cost. Shipping is free.
MEL Chemistry Box Review
Did My Son Enjoy the Science Experiments?
My 11-year-old son very much enjoyed the MEL Chemistry box. Of every type of science out there, chemistry is perhaps the subject he has had the least experience in.
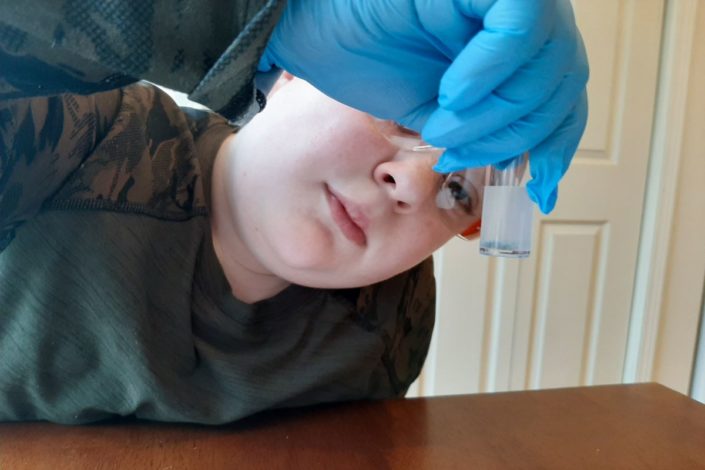
He was delighted to open the boxes and find real test tubes, Petri dishes, and chemicals. He immediately put on his safety goggles and gloves and got right to work.
MEL Science Special Offers
Pay for 9 months, get 12 months of science: MEL Science
MEL Chemistry Experiments
The experiments were fascinating, and he was able to perform both without any assistance, save for a bit of help opening bottles (it was a bit tricky with gloves).
This first box featured two experiments, both centered on tin.
Up to 3 months free with prepaid plans: MEL Science
Tin Hedgehog:
The first experiment we did was rated by MEL as 1/3 for difficulty and 2/3 for danger, presumably because it involves handling chemicals.
Protective gloves and eyewear were required.
The activity involved: transforming a small zinc pellet into a spiky pellet resembling a hedgehog.
First, my son mixed tin chloride with sodium bisulfate. Then, he poured it all into a test tube.
Finally, he submerged a tiny pellet of zinc into the tin-salt solution and watched the change that occurred: After a brief time, the smooth surface turned spiky!
Up to 3 months free with prepaid plans: MEL Science
The explanation for this was included in the experiment, in language easy for a tween to understand:
Metals are made of positively charged particles embedded in a negatively charged cloud of electrons. Like some other metals, tin would rather “chill inside a cloud of electrons,” while zinc would rather float on its own as a charged particle.
Adding the zinc pellet to the tin solution means the tin ions prompt the zinc to venture out into the solution, creating the spiky hedgehog appearance (which is technically a “crystal structure”).
Up to 3 months free with prepaid plans: MEL Science
Tin Dendrite:
The tin dendrite experiment was also rated 1/3 for difficulty and 2/3 for danger and required protective gloves and eyewear. There was an added caution note regarding the proper handling of batteries.
This experiment started off in a similar fashion:
Up to 3 months free with prepaid plans: MEL Science
My son mixed tin chloride with sodium bisulfate. But this time, he poured the solution into a petri dish and then added a couple of drops of liquid soap (provided) to make the liquid distribute more evenly.
Then, he put the batteries (our own) into the battery holder, put the wire electrodes in the solution, and then connected them to the battery holder and watched the results.
The first time my son did this, we didn’t notice much. I realized he had accidentally spilled some liquid onto the safety tray (provided), and there wasn’t enough left.
Thankfully, they provided extra! So he mixed up another batch. This time, he watched in awe as little metal “trees” appeared in the liquid.
The explanation for this likened the batteries to “electron pumps,” which suck electrons in with their positive polarity and pump them out from their negative polarity. The wires were compared with “electric hoses.”
Because the tin ions had two electrons missing, they happily accepted more electrons and turn into metallic tin; plus, the electrode (made of iron) gives some electrons to the wire, which leaves iron ions floating in the solution.
He greatly enjoyed both activities and marveled over the way a simple liquid could greatly transform a metal. He greatly enjoyed both activities and marveled over the way a simple liquid could greatly transform a metal. I think this is one of the neatest chemistry kits for 10- or 11-year-olds I have come across.
Up to 3 months free with prepaid plans: MEL Science
Benefits of MEL Science Chemistry Experiments
MEL Science offers numerous benefits both for children who are passionate about chemistry and those who simply enjoy hands-on activities or want to learn more about science.
- The experiments are truly imaginative: Sure, you can mix up baking powder and vinegar at home. But these activities are truly inventive, letting kids grow fool’s gold, make sugar snakes, or create their own hand warmers. The kit even comes with a macro lens so kids can take close-up pics of the results.
- Kids can complete experiments (mainly) on their own: It is clear much thought has gone into the clear instructions, the safety measures, and the experiments themselves to ensure no aspect is too tricky or risky for a child.
- The scientific explanations are scientific yet clear: Each experiment comes with explanations using child-friendly language to explain why each chemical reaction is occurring and why certain elements interact the way they do. Visual symbols are even embedded in the explanations to help kids make sense of them.
- The equipment and materials are high-quality: Children get to work with real scientific equipment and use interesting chemicals and metals to perform the experiments.
- Spacing out experiments keeps kids engaged: Buying one large chemistry kit might mean kids experiment a bit and then put it away. But having new activities arrive each month keeps it fresh and interesting.
MEL Science Customer Reviews
MEL Science scored an average of 4.4 out of 5 stars from 1,808 reviews across three websites. The MEL Chemistry set specifically scored 4.3 out of 5 stars from 372 reviews online. So all of the MEL Science kids have solid ratings online.
A few comments among the reviews were:
Pros
- The experiments are interesting: Many reviewers report their kids are thoroughly engaged by the interesting hands-on projects.
- The company offers extras: Many reviewers noted they appreciate the mobile app and VR lessons that come with a subscription.
- The starter kit materials are durable: The starter kit that comes with the chemistry box contains high-quality equipment (such as Petri dishes and safety goggles) that will last.
- The directions are clear: Some reviewers noted their children are able to complete many experiments independently thanks to simple visual instructions.
- The experiments yield fun results: Reviewers appreciate that their children can really see a reaction from start to finish, including a neat ending.
Cons
- Not all materials are provided: Some reviewers note they would prefer an all-inclusive kit without the need to gather any materials, such as batteries.
- Some reviewers note shipping issues: As can be the case with any mail-order product, there are reports of late or missing boxes and billing mishaps.
Overall, parents were impressed with the experiments in the MEL Science Kits. They reported that their kids loved the activities and became immersed in science as a result of them.
MEL Science Special Offers
Pay for 9 months, get 12 months of science: MEL Science
Is MEL Science Worth It?
MEL Science is certainly worth it for anyone who has kids interested in STEM—or even kids who are not interested in STEM but have a little spare time on their hands.
That’s because the activities are designed to be as entertaining as they are educational. Even reluctant learners can get excited as they perform experiments and witness chemical reactions.
MEL Science boxes offer many benefits for young scientists. I really appreciate the clear instructions, both in written form and using visuals. My kids are able to perform experiments fairly independently.
I am also impressed at the durability of the materials and equipment. While some STEM boxes rely more on paper and cardboard, MEL Science boxes often feature keepers, such as our beaker and test tube.
The company is also generous with the materials. In fact, their website notes that they try to provide sufficient materials so a child can conduct each experiment at least twice.
Some reviewers do not appreciate the fact that they at times need to provide everyday materials to complete the experiments. Personally, I am not bothered by that, as we tend to have batteries, felt-tipped markers, and other basic supplies around the house.
The instructions also explicitly state what is needed to ensure you can prepare in advance. MEL Science has replied to reviewers who noted this, saying they are simply not allowed to ship common household items, such as batteries and hydrogen peroxide.
I get a good impression of MEL Science’s customer service based on the fact that they politely respond to negative reviews in a timely manner.
I also appreciate the peace of mind that comes with ordering a subscription. MEL Science notes that subscriptions can be canceled or paused at any time with a single click … a relief for those of us who have ever felt uncomfortably locked into a subscription we no longer wanted.
The company also keeps track of order history, so even upon reactivation (which is possible anytime), your child will not receive a repeat of a previous box.
Whether your kids love science already or you just want to get them away from their screens for a bit, I wholeheartedly recommend MEL Science kits to families with children of all ages.
MEL Science Special Offers
Save up to $104.70 with prepaid plans: MEL Science
MEL Science FAQs
What age is MEL Science for?
MEL Science boxes are for children aged 5-14+ depending on the line. Their MEL Kids range is for younger children aged 5-10 years old. MEL Physics boxes are for slightly older children aged 8-14. MEL Chemistry is for older children and teenagers aged 10 and up.
How do I cancel my Mel Science subscription?
You can cancel the subscription easily from your online account with one click.
How do you use MEL Chemistry VR?
A virtual reality headset comes with your first MEL Science box. You download the MEL app to your phone or tablet. From here, you can add science lessons to be displayed on the VR set. Each one is about 3-7 minutes long. It explains chemistry concepts and asks children interactive questions.
MEL Science Special Offers
Save up to $104.70 with prepaid plans: MEL Science